It is common to see DNA damage in our cells and is rectified easily by our bodies when we are young but it becomes harder with age and leads to symptoms of aging.
MIT has carried out a new study and discovered that revitalizing a specific enzyme boosts repair of DNA damage in neurons which aids Alzheimer’s patients and others with cognitive decline. The studies before this one monitored the enzyme HDAC1 and saw how it helped repair DNA in neurons but in this study, the experts looked at what happens when it doesn’t carry out its job.
They engineered mice to be lacking in HDAC1 and studied its health compared to mice that had the enzyme. While things looked similar in their youth but with age the decline became more obvious.
ALSO READ
Too Much Work Can Kill You: Study
They saw that the mice without the HDAC1 had larger amounts of DNA damage in their neurons with age, this led to diminished synaptic plasticity. Resulting in poor performances during memory and spatial navigation tests.
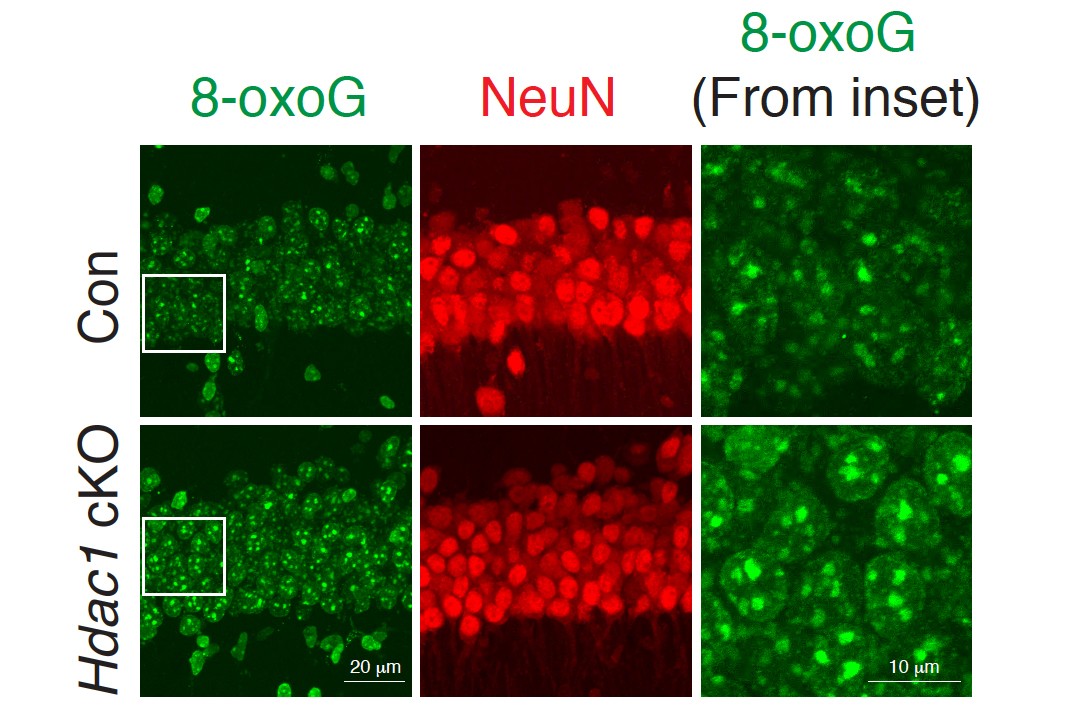
On a closer study, it was found that the mice had 8-oxo-guanine lesions, a particular type of DNA damage caused by oxidation. High levels of this are seen in people with Alzheimer’s.
Senior author of the study, Li-Huei Tsai said, “It seems that HDAC1 is really an anti-aging molecule”. “I think this is a very broadly applicable basic biology finding, because nearly all of the human neurodegenerative diseases only happen during aging. I would speculate that activating HDAC1 is beneficial in many conditions.”
For further study, they tried to treat it using exifone, an old dementia drug, which revitalizes HDAC1 and when used on mice with Alzheimer’s and healthy old mice, it was found in all the cases that it led to reduction in DNA damage to the brain and improved cognitive functions like memory.
“This study really positions HDAC1 as a potential new drug target for age-related phenotypes, as well as neurodegeneration-associated pathology and phenotypes,” says Tsai.
The research was can be read in the journal Nature Communications.