The Provincial Drug Control Authority (PDCU) has issued a public notice to recall the specific batches of a high blood pressure medicine over adulteration.
The authority has directed all the pharmacies, medical stores, wholesalers, distributors, and health facilities to remove certain batches of 2.5 mg Byscard (Nebivolol) from the shelves and stop its dispensation immediately.
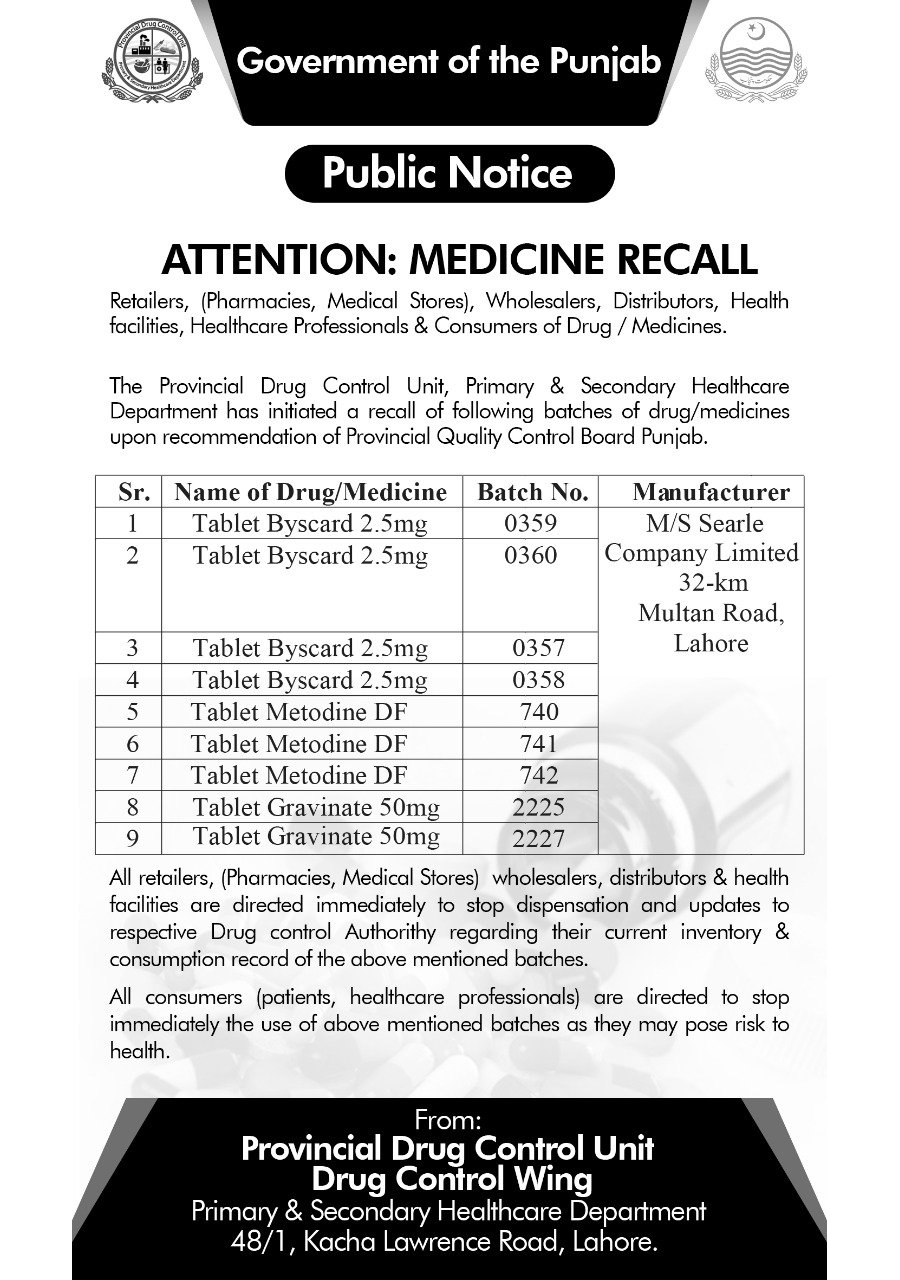
Two batches of 2.5 Byscard, i.e., 1059, 0360 are said to be adulterated with metronidazole and diloxanide. Several batches of the said tablet were dispatched for routine analysis at the drug testing laboratory in Rawalpindi.
ALSO READ
DRAP Issues Recall Alert for Critical Heart Medicine Due to Impurity
During the test, the two aforementioned batches did not respond to the analytical method of the manufacturer. This was followed by an out of specification investigation.
Other recalled medicines include Metodine DF and Gravinate 50 mg.
It has further asked the consumers and patients to stop using the said batches of medicine, as they may pose risk to health. Byscard is used to treat Hypertension and is generally prescribed by many physicians.















this is 2 weeks old news