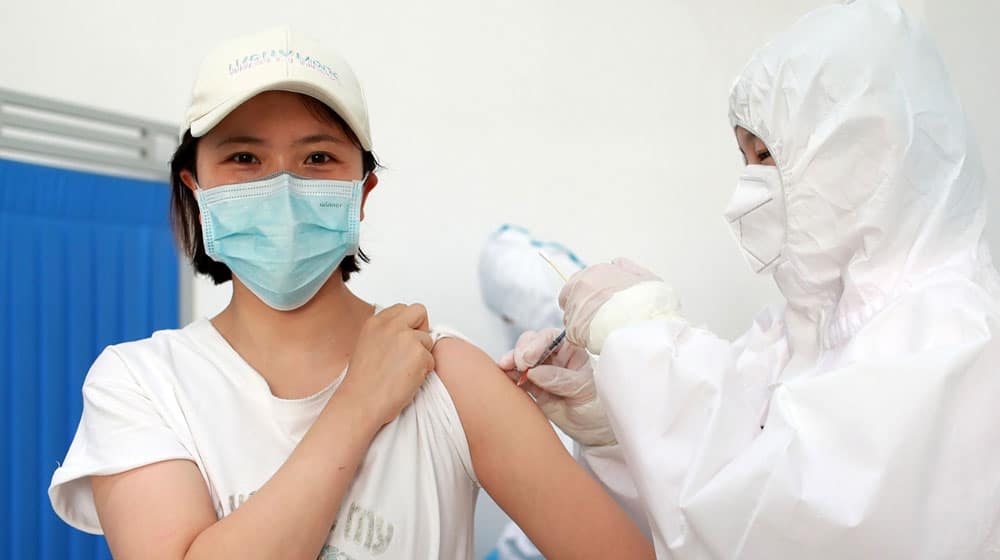
A top official with the Chinese Center for Disease Control and Prevention (CDC) has said that their coronavirus vaccines are in final stages and may be available for the public as early as November.
China is working on four Covid-19 vaccines, all under phase 3 trials. Three of these have been offered to essential workers under an emergency program.
ALSO READ
PCB Announces Covid-19 Protocols For 2020-21 Domestic Season
During an interview with the state TV, CDC Chief Guizhen Wu said that the clinical trials are running smoothly, and the vaccines could be available for commercial use by November or December.
Wu told the host that she did not experience any adverse symptoms when the trial vaccine was administered to her in April. She, however, did not mention as to which vaccine she was referring to.
ALSO READ
Pakistan Welcomes China’s New Data Security Initiative
Three of the vaccines are being developed by the state pharmaceutical giant China National Pharmaceutical Group (Sinopharm) and US-listed Sinovac Biotech. Sinopharm had stated in July that its vaccine will be ready for public use by the end of this year.
The fourth vaccine is being prepared by CanSino Biologics and was approved for military use in June. CanSino vaccine clinical trials are also being conducted in Pakistan.