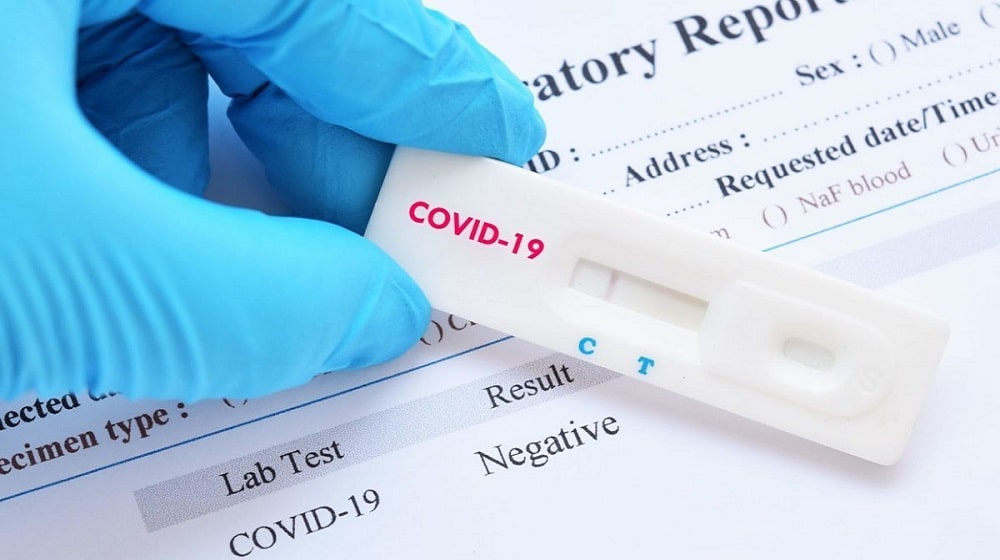
Drug Regulatory Authority of Pakistan (DRAP) has authorized an international pharmaceutical company to import a Coronavirus rapid testing kit which gives diagnosis results within 20 minutes.
According to CEO DRAP, Asim Rauf, the Coronavirus rapid testing kit has been developed in Germany.
ALSO READ
Dr. Sania Nishtar Included in 100 Most Influential Women of 2020
He added that patients will be charged Rs. 2,000 each for getting tested with the rapid testing kit.
These kits will be provided to healthcare specialists across the country who will test the patients after collecting nasal swab samples.
ALSO READ
25 November Will Be Celebrated as Pakistan Strategy Day Across the World: WEF
CEO DRAP said that the Coronavirus rapid testing kits will not be used for screening blood samples.
He termed the registration of the rapid testing kit as a tremendous success for the country as it will help in early diagnosis and timely treatment of Coronavirus patients.