The Drug Regulatory Authority of Pakistan (DRAP) has ordered the country’s pharmaceutical businesses to ensure the use of pharmaceutical-grade ingredients that have been acquired from certified suppliers.
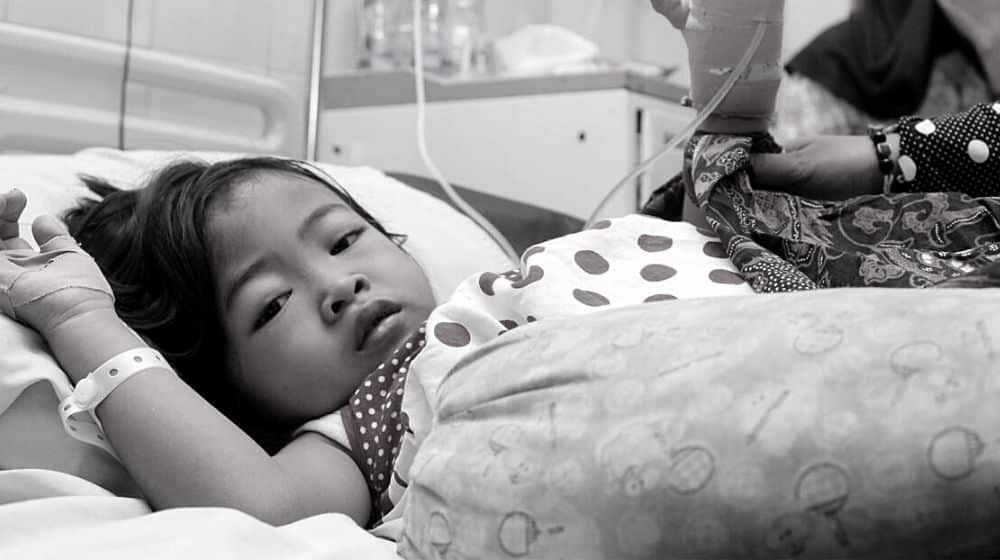
The development comes after Indian-made cough syrups killed 133 children in Indonesia and 66 in the Gambia, and caused acute kidney injuries (AKI) to hundreds of others in both countries.
According to the DRAP officials, such cases have not surfaced in Pakistan because of the increased monitoring of the drugs’ manufacturing process and assured that the syrups do not contain any harmful components.
Furthermore, the deaths of children in the above-mentioned countries were caused by the abundant presence of poisonous chemicals, diethylene glycol (DEG) and ethylene glycol (EG), in the cough syrups, a DRAP official added.
Earlier this month, the World Health Organization (WHO) issued an alert and warned that over four cough and cold syrups made by India’s Maiden Pharmaceuticals have been linked with AKI and the deaths of 66 children in the Gambia.
After this, Indonesian authorities also reported the deaths of 99 children (now 133) due to AKI caused by cough syrups. However, they did not reveal their manufacturer.
In this regard, DRAP’s alert has recommended performing a test/analysis of raw materials used in making the medicines to check for the presence of any poisonous contaminants, stressing that the pharmaceutical industry is legally obligated to provide safe medications.
In addition, it warned them not to use solvents in syrups without testing them for DEG and EG contaminants.
Besides, DRAP has directed the provincial Drug Inspectors to conduct risk-based sampling of the drugs, particularly cough syrups, and regularly report to its Quality Assurance and Lab Testing (QA and LT) division.