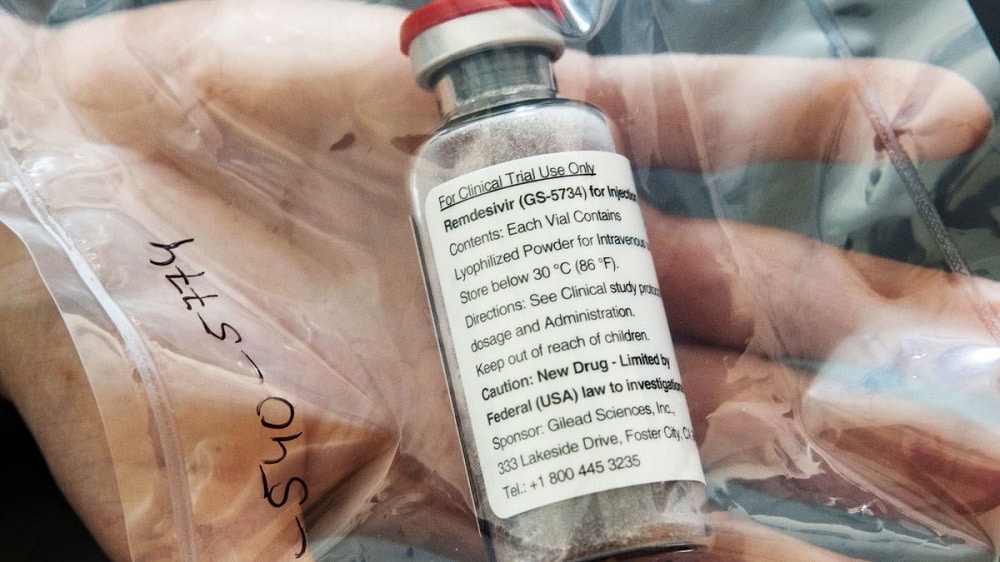
AGP Ltd, a Pakistani pharmaceutical company is planning to sell a COVID-19 treatment drug named remdesivir within one to two months in the country, reported Bloomberg.
AGP’s Chief Executive Officer Nusrat Munshi told Bloomberg that they plan to seek permission from the country’s drug authority to import the medicine.
It has a similar arrangement for treating Hepatitis.
The global push to develop coronavirus treatments and vaccines is speeding up as governments look for ways to ease lockdowns safely and restart economies.
This development comes a few weeks after remdesivir’s original producer Gilead Sciences Inc., which originally developed the drug in 2010 to treat Ebola, signed agreements with four generic drug manufacturers in India, including Mylan’s unit in the country, and one in Pakistan to accelerate development and assist in meeting the expected demand.
Ferozsons Laboratories (FEROZ) through its subsidiary BF Biosciences had successfully concluded a non-exclusive license agreement with Gilead Sciences for the manufacturing of the same drug.
AGP is Mylan’s exclusive distributor in Pakistan.
The Searle Company Limited announced last week that it had successfully entered into an exclusive licensing and marketing agreement with Beximco Pharmaceuticals, Bangladesh to supply Remdesivir in Pakistan.
“The concept of a lockdown is that it gives the country’s health-care system time to prepare,” said Munshi, adding that health-care facilities including beds haven’t been ramped up and mass testing hasn’t been conducted.
According to Munshi, AGP has also sold about 100,000 anti-body test kits since last month to hospitals and private companies looking to test their staff as more people return to work. The company has another 100,000 kits and is considering importing more as it is seeing high demand.