The US Food and Drug Administration (FDA) has granted Emergency Use Authorization (EUA) to the first Coronavirus self-diagnostic molecular test that not only can be performed at home but it also delivers accurate results in 30 minutes.
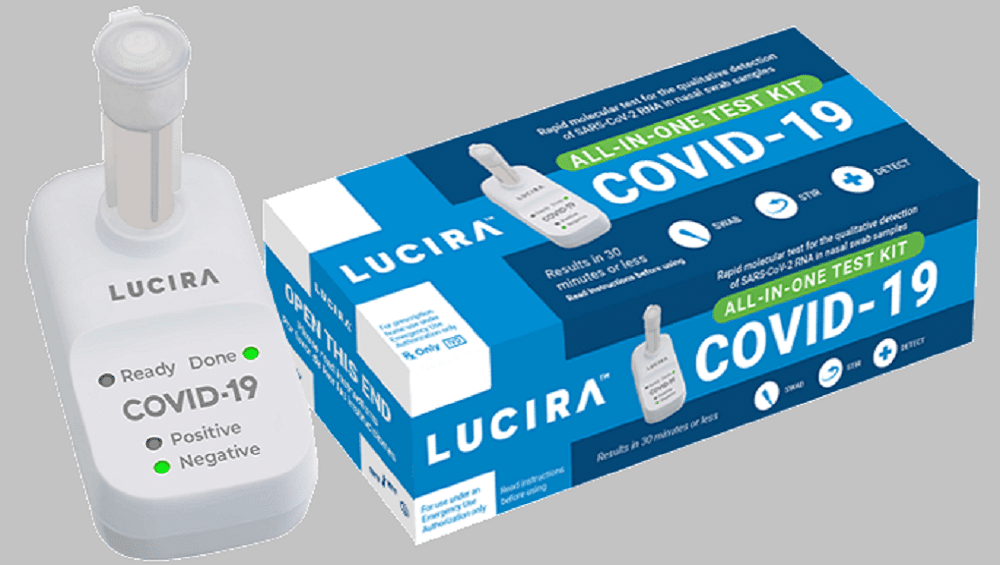
According to details, the single-use test kit has been designed and developed by Lucira Health, a California-based biotechnology company.
ALSO READ
Pfizer Becomes the Most Effective COVID-19 Vaccine Based on Trial Reports
In an official statement, FDA Commissioner, Stephen Hahn, has said the action underscores FDA’s ongoing commitment to expand access to COVID-19 testing.
This new testing option is an important diagnostic advancement to address the pandemic and reduce the public burden of disease transmission.
CEO Lucira Health, Erik Engelson, has said:
Molecular tests like Lucira’s are 50 to 60 times more sensitive than antigen tests and are considered the ‘gold standard’ for determining if someone is infected.
Besides self-diagnosis, Lucira’s testing kit can be used in point-of-care (POC) settings such as doctor’s offices, hospitals, urgent care centers, and emergency rooms for all ages.
Lucira Health’s self-diagnostic testing kit will be widely available by March 2021 and will be priced at $50.
ALSO READ
Smart Lockdown Imposed in Punjab’s Six Major Cities Amid Rising COVID-19 Cases
How Does it Work?
The test works by swirling the self-collected nasal swab sample in a vial that is then placed in the testing kit.
Within 30 minutes, the result can be read directly from the kit’s light-up display that shows whether an individual is positive or negative for the SARS-CoV-2, the Coronavirus which causes the COVID-19 disease.
FDA has advised individuals with positive results, which indicates the presence of SARS-CoV-2, to self-isolate and seek additional care from their health care provider.















I want to order it.Where we can purchase it in Pakistan?
When the PCR test itself is a farce and the inventor of the test (Kary Mullis) has repeatedly stated its not meant to be used as a diagnostic tool, what’s the point of releasing a self-testing device? more profit?
My personal favorite article that shows how ridiculous the Covid test is the singer Erykah Badu (search for it). Her right nostril gave a negative result, left nostril gave positive :/