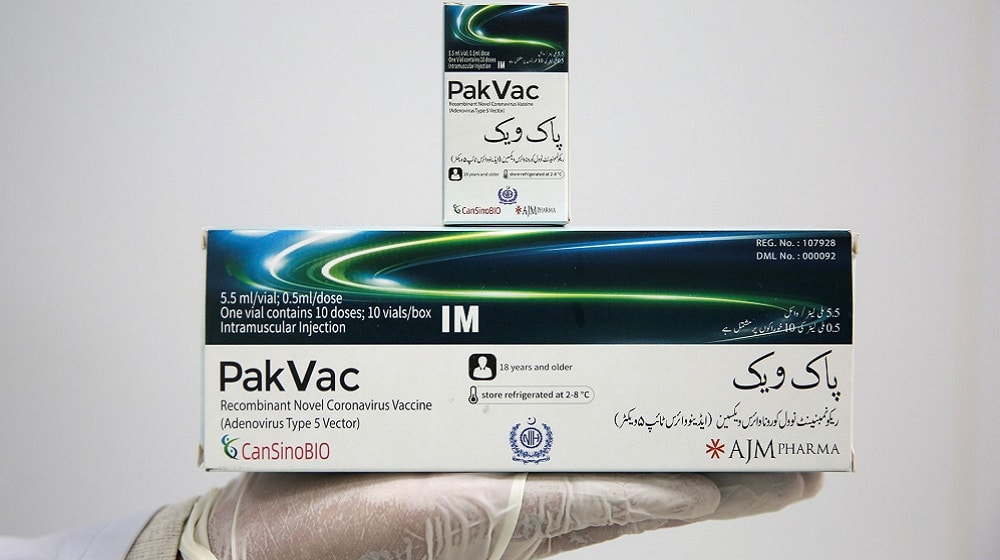
PakVac, Pakistan’s first locally-packaged single-dose COVID-19 vaccine, is acceptable in the US and UK, SAPM on Health, Dr. Faisal Sultan, has revealed.
While speaking in a talk show, Dr. Faisal Sultan clarified that PakVac is acceptable in all those countries, including the US and UK, that do not require inbound travelers to be vaccinated with certain Coronavirus vaccines.
ALSO READ
CM KP Suspends Officials of Public Offices in Surprise Visits
Also known as Ad5-nCoV, PakVac is a rebranded version of a Chinese single-dose Coronavirus vaccine that was originally developed by CanSinoBIO in collaboration with the Beijing Institute of Biotechnology.
The vaccine was tested in large-scale phase-III clinical trials that were held in five countries, including Pakistan, in which more than 40,000 volunteers participated.
Results of trials were published in February this year which confirmed that the CanSinoBIO vaccine was 65% effective at preventing symptomatic cases and 91% effective at preventing severe infection. As for the Pakistani subset, the vaccine offered 75% protection against symptomatic cases and 100% protection against severe infection.
ALSO READ
Ceiling of New Islamabad Airport Collapses
In May, the National Institute of Health (NIH) Islamabad launched the local packaging of PakVac after its raw material reached Pakistan from China.
NIH is preparing 3 million doses of PakVac each month that has significantly reduced Pakistan’s dependence on imported COVID-19 vaccines and ensure the sufficient availability of the Coronavirus vaccine.