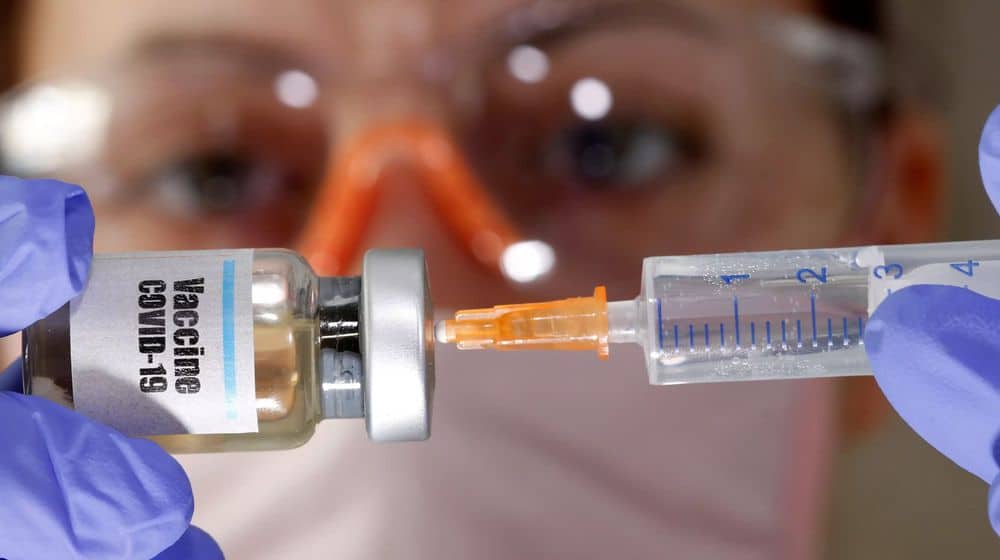
Vice-Chancellor (VC) Health Services Academy (HSA) and Chairman National Vaccine Committee, Dr. Asad Hafeez, has revealed that HSA is devising a transparent mechanism that would ensure countrywide availability of any Coronavirus vaccine that receives international regulatory approval.
Under the said mechanism, any Covid-19 vaccine that becomes available internationally would be distributed at both provincial and district levels within days after registering it with the Drug Regulatory Authority of Pakistan (DRAP). The vaccine would be delivered to frontline healthcare workers and vulnerable populations on a priority basis.
ALSO READ
All You Need to Know About MG Motors & Expected Car Line-up for Pakistan
While addressing a national-level consultative meeting organized by the Ministry of National Health Services (NHS), HSA, and Health and Population Think Tank, Dr. Hafeez said that 6 Coronavirus vaccines are currently being evaluated around the world in the Phase-III clinical trials.
He added that arrangements, on the recommendation of the National Command Operation Center (NCOC), are underway as well to ensure adequate cold storage facilities for the Coronavirus vaccine on par with global safety standards.
ALSO READ
Pakistan Secures Landmark Decision in $6 Billion Reko Diq Case
Special Assistant to the Prime Minister (SAPM) on NHS, Dr. Faisal Sultan, said that the federal government would extend maximum support to any effort directed at bringing the vaccine to the country.
Officials of both federal and provincial health departments, Ministry of Science and Technology, Ministry of Federal Education, NCOC, DRAP, and healthcare experts from the National Institute of Health (NIH) and the Aga Khan University also attended the meeting.