In a statement on Friday, the Drug Regulatory Authority of Pakistan (DRAP) said that it has issued a recall of certain batches of cough syrups due to suspected contamination with diethylene glycol (DEG) and ethylene glycol (EG).
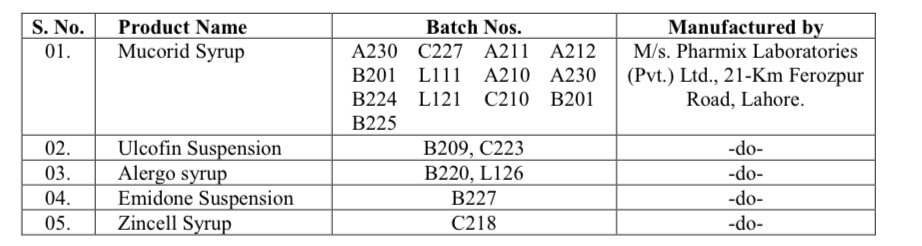
The World Health Organization (WHO) approached DRAP and informed it about the suspected presence of DEG/EG impurities in batch No. B220 of Alergo Syrup, identified in the Maldives, which was manufactured by M/s. Pharmix Laboratories (Pvt.) Ltd., Lahore.
After getting information from WHO, the drug regulatory authority decided to launch an investigation into the matter. It reached the conclusion that the impurities might also be present in other batches and products.
Following are the details about the products and batches:
DRAP added that they have taken the step as a ‘precautionary measure’, aimed at safeguarding the health of the public against the potentially harmful effects of these impurities.
It should be noted that in the past, syrups meant for children contaminated with high levels of DEG and EG led to severe adverse events and fatalities in a few countries.
DRAP has ordered the Lahore-based manufacturing company to immediately recall the above-mentioned products before they cause any harm.
Furthermore, all the pharmacists and chemists working at distribution and pharmacies have been advised against the supply of these products.
The general public has also been advised to stop using this product bearing the affected batch numbers. They have been asked to contact their doctor in case of any complications due to the usage of the product.