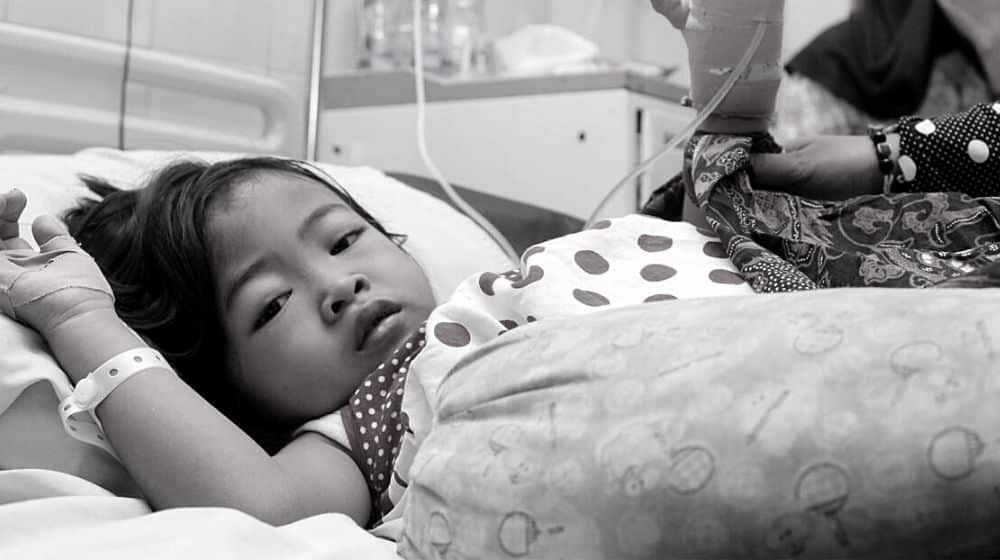
Several cough syrups manufactured by an Indian pharmaceutical company have been linked with acute kidney injuries (AKI) and the deaths of 99 children in Indonesia this year.
Indonesia’s Health Minister has said that apart from the 99 deaths, another 206 children are in serious condition due to AKI. However, the Indonesian authorities have not revealed the company’s name or the type of syrups linked with the deaths and kidney disease.
As of now, Indonesia has imposed a comprehensive ban on all syrups and other liquid drugs in the country. According to health officials, the 206 children affected by AKI are aged under five.
Earlier this month, the World Health Organization (WHO) issued an alert and warned that over four cough and cold syrups made by India’s Maiden Pharmaceuticals have been linked with AKI and the deaths of 66 children in the Gambia.
The four medicines include Promethazine Oral Solution, Kofexmalin Baby Cough Syrup, Makoff Baby Cough Syrup, and Magrip N Cold Syrup, as stated in the WHO’s alert.
In addition, the WHO also warned of global exposure to contaminated medicines due to their export outside the African country.





















Shame on the Company, and regulations of the country, should take action and banned this company soon, Health is important for all, without health we can afford any kind of work.
so, health is wealth, and please take a look at health insurance for your family and babies